If you follow a lot of tech circles, you may have seen graphene (a super-thin layer of carbon arranged in such a way that it has electrical properties verging on miraculous) come up in the news quite a bit, receiving plaudits about its massively fluid electrical conductivity and possible applications in several different technologies. What you haven’t heard much of is the ugly part of graphene: It’s impossible to build semiconductor transistors out of the material as it stands now since it has no electrical band gap to speak of. If that sounds confusing, that’s alright. That’s what this article is for!
Band Gap? What’s That?
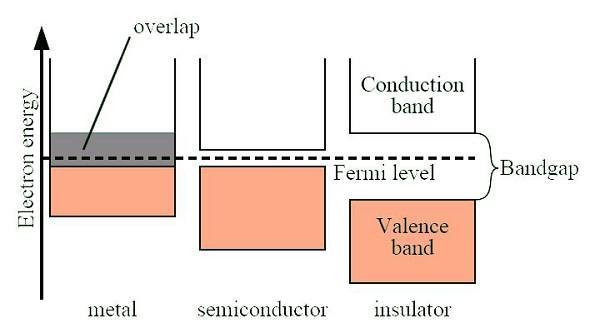
A band gap is a tiny space in between a conduction band and a valence band that tells us at what level current will actually flow between the two. It’s like a little gatekeeper that keeps an electrical charge in one space until it is “turned off.” Virtually all chips on computers are made of a semiconductor material, which means that it has a moderate band gap that makes it neither conduct electricity so readily nor reject every electrical charge. This has to do with basic molecular structure, so there is quite a bit of chemistry involved in building a chip.
Very large band gaps exist in materials like rubber which will resist electrical currents so much that it would much rather catch fire than retain the charge. That’s why you use rubber to insulate the wires inside of cables. Materials with a negligible band gap are known as conductors, while those with virtually no band gap whatsoever are known as superconductors.
Today most chips are made of silicon, which serves as a very sturdy and reliable semiconductor. Remember, we need semiconductors that can quickly be turned on and off at will, not superconductors, which will lose the charge they were given the moment the band no longer supplies it.
Why Is Graphene Not Good for Building Chips?

As I mentioned earlier, graphene is an extremely efficient conductor of electricity but nothing much more than that. It can push a charge at an incredible speed, but it cannot retain it. In a binary system you may need to retain data so that your running programs don’t just close the instant they open. It’s important in a RAM chip, for example, to ensure that the data inside it can stay put and remain readable for the foreseeable future. When a transistor is in the “on” state, it registers a “1.” In an “off” state, it registers a “0.” A superconductor would be unable to “switch off” because the difference between “on” and “off” voltage is so small (because of the tiny band gap I mentioned earlier).
That’s not to say that graphene wouldn’t have a place in a modern-day computer. It certainly could be used to deliver information from one point to another quickly. Also, if supplemented by other technology, we could possibly see graphene used in transistors at some point in the future. Whether that would be an efficient investment of capital is up to the industry to decide.
There’s Another Material!
One of the problems with silicon is its inflexibility when working on ultra-thin surfaces. A piece of silicon could only be shaved so thin for it to be functional. That’s why we were exploring the use of graphene in the first place (it’s one single atom thick). Since graphene may not prove promising without investing truckloads of money into its development, scientists began trying other materials, one of which is titanium trisulfide (TiS3). The material not only has the ability to function even at the thickness of a single molecule, but it also has a band gap very similar to that of silicon.
The implications of this are far-reaching for miniature technology products which pack a vast amount of hardware in a very constrained amount of space. Thinner materials will also dissipate heat more efficiently, making them favorable for large power-hungry computers.
It’s now your turn to share your input on the quest to replace silicon. Leave a comment below with your thoughts!







